INTRO
In episode 65 of the Japan Distilled podcast, we discuss something that is often overlooked in discussions of distilled spirits production. What’s happening inside the pot still?
CREDITS
Theme Song: Begin Anywhere by Tomoko Miyata (http://tomokomiyata.net/)
Mixing and Editing: Rich Pav (https://www.uncannyrobotpodcast.com/)
HOSTS
CHRISTOPHER PELLEGRINI Vermont born and bred, long-time Tokyo resident and author of The Shochu Handbook, Christopher learned about delicious fermentations as a beer brewer at Otter Creek (Middlebury, VT). He now spends most of his waking hours convincing strangers that shochu and awamori are unlike anything they’ve ever tried before.
STEPHEN LYMAN discovered Japan’s indigenous spirits at an izakaya in New York City. He was so enthralled that he now lives in Japan and works in a tiny craft shochu distillery every autumn. His first book, The Complete Guide to Japanese Drinks, was nominated for a 2020 James Beard Award.
Stephen and Christopher sometimes pretend to be chemists even though they are not.
If you have any comments or questions about this episode, please reach out to Stephen or Christopher via Twitter. We would love to hear from you.
SHOW NOTES
The Pot Still
When it comes to distilled beverage alcohol, there are two primary still designs. The pot still and the column still. A column still is what’s used to turn crude oil into high-test gasoline and that’s about as much as you need to know about how its used to make beverage alcohol. Think vodka, soju, and most budget minded gins, rums, and whiskies.
The pot still is where things get interesting (and more expensive). The fermentation is boiled and evaporated out of the still one time for each run. In this batched process, the alcohol yields are lower, but much more interesting flavors and aromas are retained. Think Jamaican pot still rum, single malt Scotch, mezcal, and, of course, Honkaku Shochu and Ryukyu Awamori.

What’s Happening Inside the Pot Still?
This is where things get endlessly complex, confusing, and interesting. This is also where many people decide to just enjoy the beverages rather than understanding their production process in a deeper way. And that’s okay.
A fermentation, whether it be a “simple” rum molasses fermentation, a “beer” for whisky making, or a porridge-like sweet potato shochu fermentation, is a complex community of living organic matter, dead organic matter, liquids, solids, sugars, starches, proteins, fats, and myriad other things.
Each and every one of these things has a vapor point, or the temperature at which is will convert from liquid form to gaseous form. This is what will determine when that particular compound will escape through the neck of the pot still only to be re-liquified in a cooling apparatus to become part of the resulting spirit.
As you may recall from high school chemistry class, the boiling point (vapor point) for water is 100 degrees Celsius. For ethanol (the “goal” of beverage alcohol distillation), it is around 78.4 degrees Celsius. Therefore, to extract the spirit from the fermentation, you want the temperature inside the pot still to be somewhere between 78.4 and 100 degrees Celsius.
This is not to say that anywhere between those two temperatures is fine. It’s not. All of the other compounds inside the pot still will be evaporating or not depending on the temperature and this can change the flavor and aroma of a spirit in profound ways. And this is where the art comes in. The distiller who knows what she’s doing will optimize the temperature inside the still to capture the maximum ethanol yield in perfect harmony with the ideal flavor and aroma profile for what they are trying to make. That’s where the magic lies. Everything else is just chemistry.
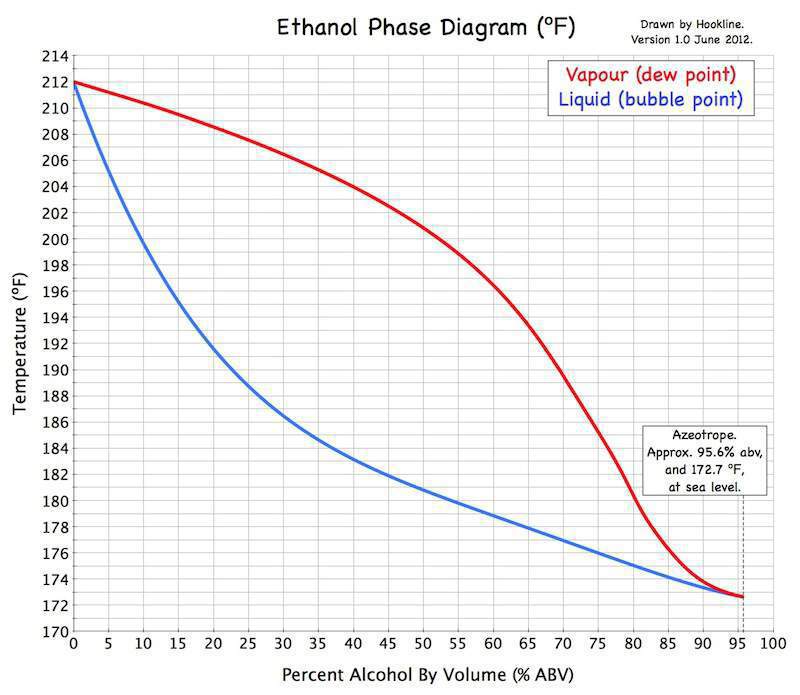
Just Scratching the Surface
To learn more, check out the Moonshine Distiller website, which is chock full of some of the best English language descriptions about what’s happening inside the pot still.

Leave a Reply